Your cart is currently empty!
Have confidence in your labeling reaction, save time and money
Imagine planning a time-consuming experiment with expensive antibodies that should be conjugated to your label of choice, only to have the experiment fail with no clear reason. Is it possible your antibody wasn’t even labeled to begin with?
Antibodies are expensive and your time is valuable; therefore, you should have absolute confidence in your antibody labeling reactions prior to conducting downstream applications. The problem is, effectively checking whether your antibody conjugation was successful prior to its use can be difficult and therefore is rarely done. As a result, a great deal of uncertainty can stem from these ambiguous antibody-conjugates, leaving researchers to question whether their antibody labeling reactions are a possible culprit when a crucial experiment fails.
Commercially available antibody labeling kits offer inadequate solutions for confirming conjugations, with their availability limited to only a few labels such as specific fluorophores, Biotin, and HRP. These kits often come at an extra cost to researchers, take additional time to perform the conjugation-confirmation experiment, and may result in unclear conclusions due to low accuracy. A more confident approach to antibody conjugations is necessary for researchers to trust their labeling reactions.
There is a solution: oYo-Link® instils confidence in your labeling reaction!
Easily confirm that oYo-Link® has successfully attached your label to your antibody via SDS-PAGE

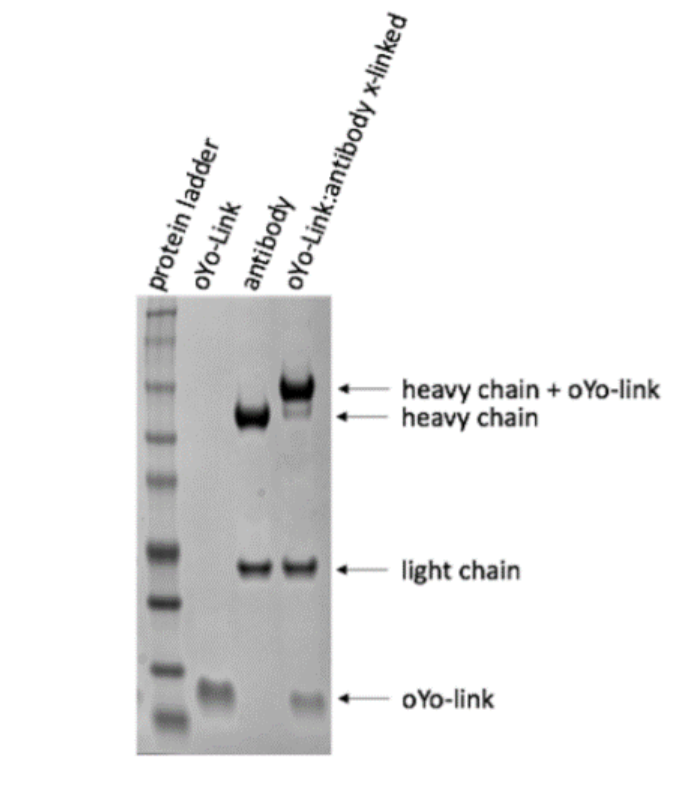
AlphaThera’s oYo-Link antibody labeling reagents allow scientists to easily confirm the successful conjugation of their antibody to a range of labels by simply performing SDS-PAGE. Competing antibody labeling reactions based on traditional chemistries that conjugate small labels, e.g. dyes, biotin, drugs, are too small to cause gel shift, making it difficult to visualize their success via SDS-PAGE. Unlike these competing antibody labeling products that may be difficult to detect, oYo-Link consists of a small (8 kDa) photoreactive antibody-binding domain that site-specifically and covalently labels the heavy chain of antibodies upon Illumination with a black light. Successful photocrosslinking leads to a slight increase in the molecular weight of the antibody heavy chain, which can be easily detected by a noticeable upward shift on a gel.
Gauge antibody labeling efficiency
Beyond simply checking whether an antibody is labeled or not, scientists can further use oYo-Link to assess the efficiency of their labeling reaction. Since any antibody that is not labeled will not be shifted upwards, the relative intensity of the upper band will roughly indicate how much antibody has been efficiently labeled.
Avoid wasting antibody
As little as 1 ug of antibody can be seen via SDS-PAGE, so it is not necessary to waste large amounts of material to confirm that the conjugation was successful. Moreover, oYo-Link reagents do not have to be used all at once, you can label just what is needed for your experiment and save the rest for later, preventing the waste of oYo-Link and antibody.
Running a crosslinking gel with oYo-Link to confirm successful conjugation
The efficiency of antibody labeling with oYo-Link can be determined using SDS-Page. To check successful labeling, unconjugated antibody and conjugated antibody are loaded into two different wells of a gel for SDS-PAGE and under reducing conditions. The oYo-Link-conjugated IgG heavy chain will appear as a higher molecular weight band (i.e. shifted upward) compared to the unconjugated IgG heavy chain, as shown in Figure 1.
Basic methodology
-
Load 1 µg of unconjugated antibody and 1 µg of conjugated antibody into two different wells of a gel for SDS-PAGE.
-
Check your protein gel and staining system’s recommendations for protein loading, which may deviate from 1 µg.
-
Under reducing conditions, the oYo-Link-conjugated IgG heavy chain will appear as a higher molecular weight band (i.e. shifted upward) compared to the unconjugated IgG heavy chain (as shown in Figure 1).